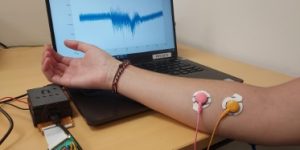
REHYB@TECNALIA: TECNALIA has developed a new EMG signal acquisition device
10.07.2023. As part of the REHYB project, TECNALIA has developed a new EMG signal acquisition device that improves the performance of the previous device integrated into the beta prototypes developed in the REHYB project.